| Gastroenterology Research, ISSN 1918-2805 print, 1918-2813 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Gastroenterol Res and Elmer Press Inc |
| Journal website https://www.gastrores.org |
Original Article
Volume 16, Number 4, August 2023, pages 209-216
Fecal Microbiota Transplantation in Human Immunodeficiency Virus-Infected Patient Population: A Systematic Review and Meta-Analysis
Adnan Malika, c, Muhammad Imran Malikb
aMountain Vista Medical Center, Mesa, AZ, USA
bHematology Specialty, Airedale General Hospital, West Yorkshire, UK
cCorresponding Author: Adnan Malik, Mountain Vista Medical Center, Mesa, AZ, USA
Manuscript submitted April 8, 2023, accepted June 22, 2023, published online August 26, 2023
Short title: FMT in HIV-Infected Patient Population
doi: https://doi.org/10.14740/gr1624
| Abstract | ▴Top |
Background: Patients with human immunodeficiency virus (HIV) infection suffer from alterations in gut microbiota due to recurrent gastrointestinal infections and systemic inflammation. Fecal microbiota transplantation (FMT) appears to be a potential therapy; however, there are concerns about its safety. Likewise, no previous meta-analysis evaluated FMT efficacy in HIV-infected patients.
Methods: We conducted a thorough electronic search on PubMed, Scopus, OVID, Web of Science, and Cochrane CENTRAL for clinical studies assessing the safety and efficacy of FMT in patients with HIV and gastrointestinal dysbiosis, where FMT was indicated to restore the disrupted microbiota.
Results: FMT significantly restored the typical microbiome in patients with Clostridium difficile (C. difficile) and non-C. difficile and reduced the risk of gastrointestinal infections in HIV patients receiving antiretroviral therapy (odds ratio (OR) = 0.774, 95% confidence interval (CI): (0.62, 0.966)). Furthermore, adverse events, such as distention and bloating, associated with FMT were comparable between HIV and health controls (OR = 0.60, 95% CI: (0.07, 4.6)), with no statistical difference.
Conclusions: Current evidence demonstrated that FMT is safe and effective in HIV patients suffering from alterations in gut microbiota. We recommend further multi-centric clinical studies to address the optimal transplant amount and source for FMT. To the best of our knowledge, this is the first meta-analysis to assess the safety and efficacy of FMT in patients with HIV.
Keywords: Human immunodeficiency virus; AIDS; Fecal transplantation; Gut microbiota; Meta-analysis
| Introduction | ▴Top |
Human immunodeficiency virus (HIV) is the causative organism of acquired immunodeficiency syndrome (AIDS). Our immune system protects against viral and bacterial infections. White blood cells (WBCs), including CD4 cells, are an immune system component [1]. HIV infects and progressively kills CD4 cells, which results in the loss of the body’s ability to recognize the infections. The late stage of HIV infection is AIDS, in which the patient usually develops opportunistic infections [2].
HIV can affect the gastrointestinal tract (GIT) in the acute stage. The lymphocytes of the GIT contain an essential receptor (chemokine receptor type 5 (CCR5)) for the entry of CCR5-tropic HIV-1 into CD4 cells. The decline in intestinal CD4 cells is more significant than in extra-intestinal lymphocytes [3]. The renewal of the immunity of GIT mucosa remains incomplete despite the use of highly active antiretroviral therapy (HAART). Disruption of GIT mucosa leads to microbial translocation [4].
GIT symptoms of HIV infection include nausea, vomiting, diarrhea, tenesmus, abdominal pain, dysphagia, and odynophagia [5]. HIV can cause GIT inflammation, malabsorption, and vitamin B12 deficiency without apparent viral or bacterial infection [6]. HIV-related GIT opportunistic infections can be associated with candidiasis, cytomegalovirus (CMV), Giardia, Microsporidia, Strongyloides stercoralis, Clostridia, or fungal infection (cryptococcosis, histoplasmosis) [7]. GIT contains trillions of microbes of different strains known as intestinal microbiota. These microbiotas contribute to the body’s immune system [8]. Disruption of the mucosal barrier due to the alteration of intestinal microbiota (dysbiosis) leads to microbial translocation to the system circulation [9].
Methods of gut microbiota modification include diet changes, certain drugs such as metformin, antibiotics, probiotics, and fecal microbiota transplantation (FMT). In FMT, the patient receives fecal microorganisms from a healthy donor. Previous trials reported that FMT reduced gut damage without serious side effects. The safety of FMT is still questionable due to the high variation in GIT microbiota between the healthy donor and the patient. A limited number of patients participated in studies assessing FMT in immunocompromised patients [10].
We aimed to assess the efficacy and safety of FMT as a potential therapy for HIV-related GIT symptoms.
| Materials and Methods | ▴Top |
Per the latest version of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and Cochrane Handbook for Systematic Reviews of Interventions, we prospectively determined the study objectives, search strategy, eligibility criteria, and analytical techniques for conducting this systematic review and meta-analysis [11, 12]. The Institutional Review Board (IRB) approval and ethical compliance declaration are not applicable for this study.
Search strategy
We conducted a thorough electronic search on PubMed, Scopus, OVID, Web of Science, and Cochrane CENTRAL from inception until November 2021 using the following search terms: “fecal microbiota transplantation”, “gut microbiome”, “bacteriotherapy”, “Clostridium difficile”, “FMT,” “immunocompromised”, “microbiota”, “immunosuppression”, “HIV”, or “AIDS”.
References of all retrieved articles were also screened and assessed for additional sources.
Eligibility criteria
A preliminary selection of studies was performed based on titles and abstracts. Two investigators then independently reviewed the full content of each selected study using the following inclusion criteria: 1) Clinical studies assessing the safety and efficacy of FMT in patients with HIV and gastrointestinal intestinal dysbiosis with or without Clostridium difficile infection; 2) FMT was indicated to restore the disrupted microbiota; 3) Measured complications and effectiveness outcomes; 4) Provided sufficient data for statistical analysis. Studies were excluded if they contained any one of the following exclusion criteria: 1) Essay and review articles; 2) Studies conducted on patients without HIV; 3) Studies that did not include FMT as an active intervention.
Data extraction
Two reviewers performed data extraction independently, and any disagreement was addressed by discussion and consensus. The extracted data covered: 1) The general characteristics of each study (authors; year; setting; sample size; percentage of HAART, methods and amounts of FMT delivery); 2) The outcomes measured (stool amount per transplant, donor relationship, adverse events (AEs), and final findings).
Risk of bias assessment
The risk of bias within each included study was assessed by two independent authors using the Newcastle-Ottawa Scale for cohort studies. The tool consists of eight items with three subscales and a total maximum score of 9, where scores ≥ 7 implicate a high-quality article and < 5 indicate low-quality research [13].
Data analysis
All statistical analyses were performed using Open Meta Analyst (AHRQ, CEBM; Brown University, USA) and STATA version 16.0 (StataCorp LLC, College Station, TX 77845, USA). We ultimately employed the random-effects model with the Der-Simonian Liard method [12]. Extracted data were dichotomous (events and no events) and were pooled as weighted proportions and odds ratios (RR) with 95% confidence intervals (CI) [14]. The pooled rates of proportions were calculated through the Freeman-Tukey transformation meta-analysis of proportions using MedCalc (Version 15.0; MedCalc Software, Ostend, Belgium). Heterogeneity between studies was examined visually and statistically through Chi-square and I2 tests: a Q statistic with P < 0.1 indicated heterogeneity, whereas I2 values of 0%, 25%, 50%, and 75% represented no, low, moderate, and high heterogeneity, respectively [14]. When detecting considerable heterogeneity, we performed sensitivity analyses to ascertain the source of heterogeneity by excluding one study at a time. Publication bias was visually examined through funnel plot symmetry and mathematically through Egger/s regression test, Begg’s test, and Duval’s non-parametric trim-and-fill analysis [15-17].
| Results | ▴Top |
Search results and characteristics of included studies
Our search retrieved 486 unique citations from searching electronic databases. Following the title and abstract screening, 19 full-text articles were retrieved and screened for eligibility. Of them, nine articles were excluded, and 10 studies (n = 310 patients) were reviewed in detail and included in this meta-analysis (Fig. 1, PRISMA flow diagram) [9, 18-30].
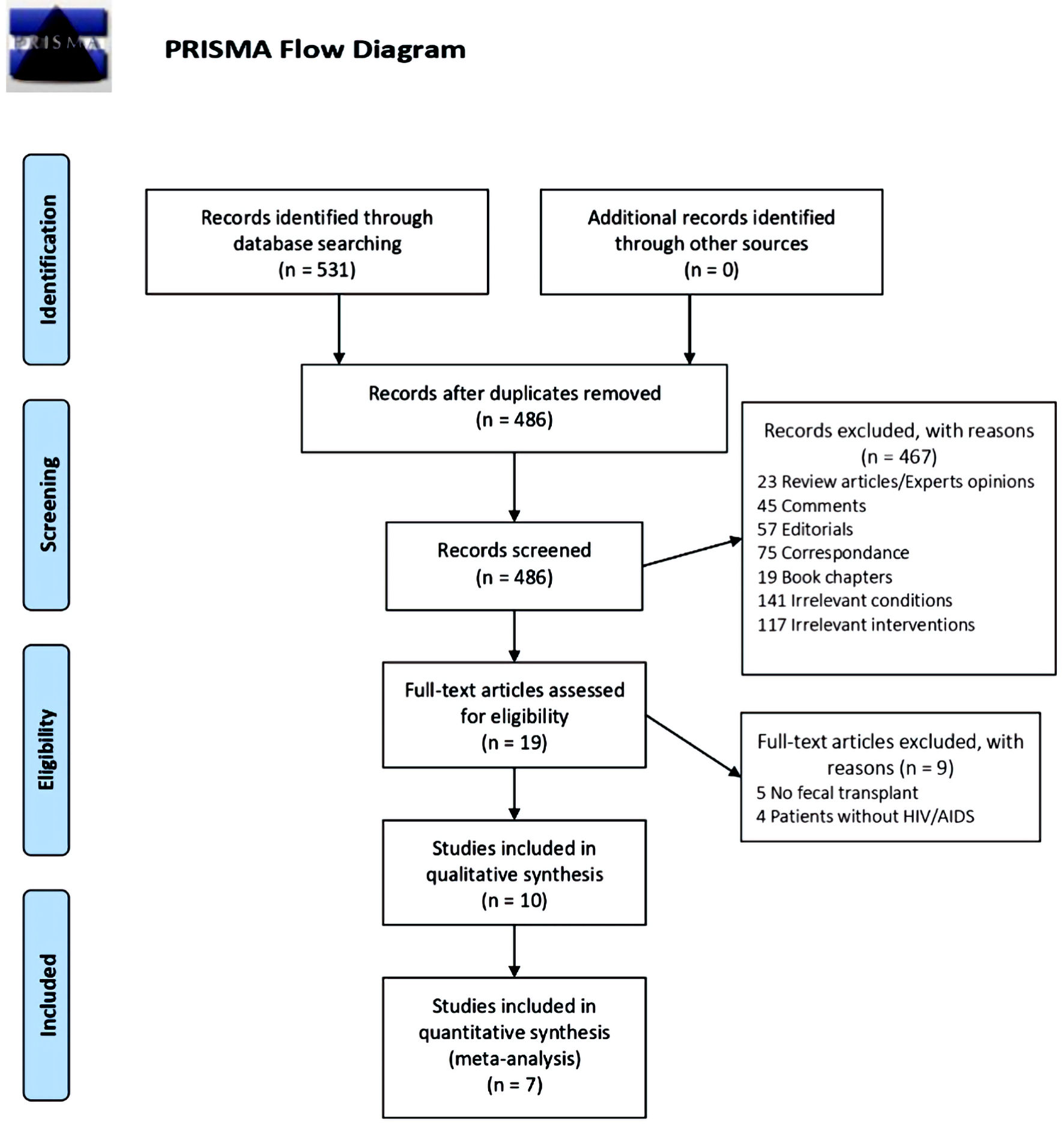 Click for large image | Figure 1. PRISMA flow diagram. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses. |
The bibliography of the included randomized controlled trials (RCT) was manually examined but added no further records. All of the included studies were conducted between 2013 and 2021. Table 1 [18-20, 23-25, 27,-30] summarizes the characteristics of included patients and studies.
 Click to view | Table 1. Characteristics and Summary of Included Studies |
The potential sources of bias
Following the Newcastle-Ottawa Scale, the quality of the included studies ranged from mild to moderate. A summary of quality assessment domains and authors’ judgments with justifications are shown here (Supplementary Material 1, www.gastrores.org). Funnel plots of the standard errors versus the effect size could not be drawn since included studies are less than 10. However, both Egger’s (P = 0.3) and Begg’s tests (P = 1.2) showed no small-study effects. Also, we engaged the trim-and-fill method to confirm the robustness of the results, which revealed no change in the effect size after imputing zero missing articles (Supplementary Material 2, www.gastrores.org).
Outcomes
Overall efficacy
FMT potentially restored the normal microbiome and reduced the risk of gastrointestinal infections in HIV patients receiving HAART (OR = 0.62, 95% CI: (0.25, 0.99)), with no statistical significance. The pooled analysis was moderately heterogeneous (I2 = 60.3%, P = 0.01) (Fig. 2). However, following the sensitivity analyses with the exclusion of the study of Mandalia et al 2016 [19], the heterogeneity resolved (I2 = 46.5%, P = 0.00), and the effect was statistically significant (OR = 0.774, 95% CI: (0.62, 0.966)).
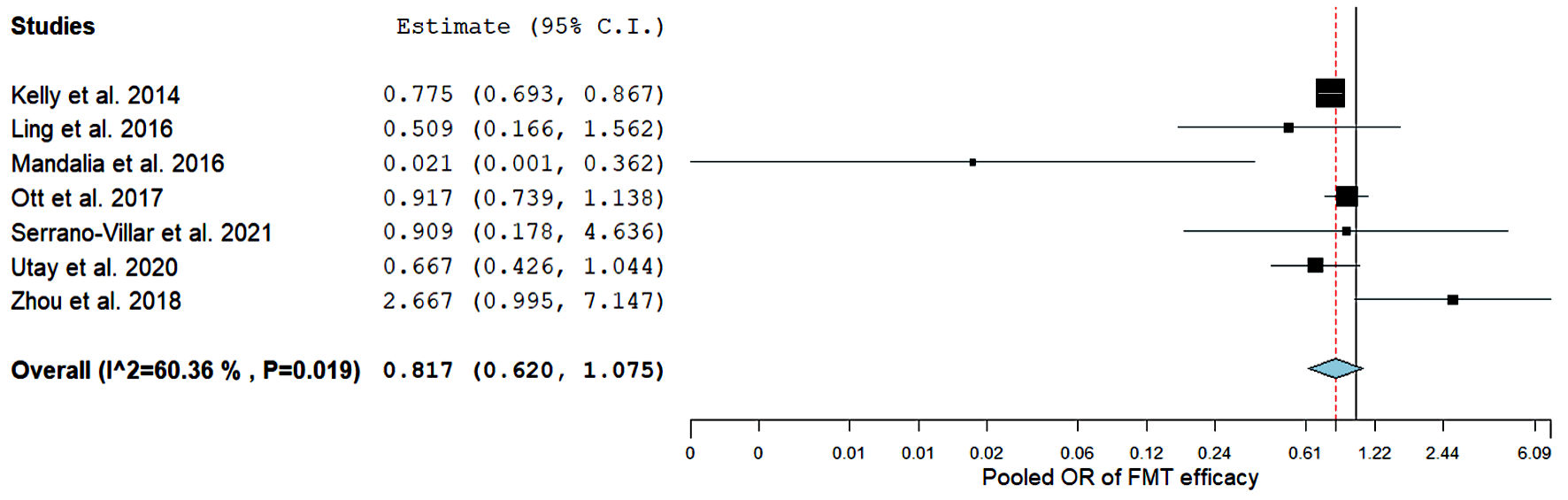 Click for large image | Figure 2. Forest plot for FMT efficacy. FMT: fecal microbiota transplantation; OR: odds ratio; CI: confidence interval. |
Overall safety
AEs, such as distention and bloating, associated with FMT were comparable between HIV and health controls (OR = 0.60, 95% CI: (0.07, 4.6)) (Fig. 3), with no statistical difference. However, the pooled analysis was substantially heterogeneous (I2 = 93.3%, P= 0.00), and further sensitivity analyses failed to resolve heterogeneity. Therefore, the random-effects model was employed.
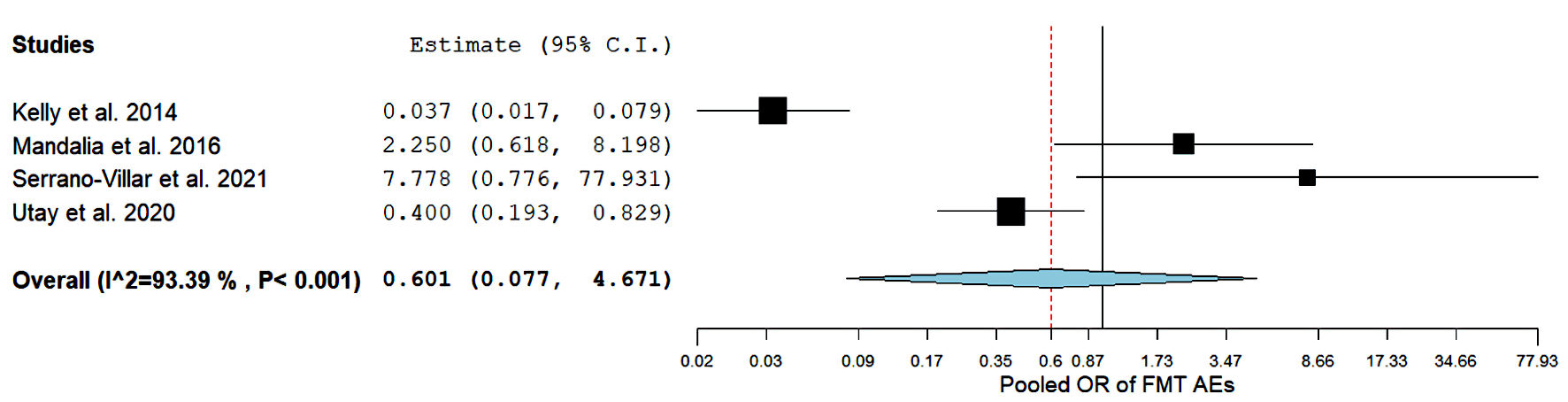 Click for large image | Figure 3. Forest plots for FMT Adverse events. FMT: fecal microbiota transplantation; AEs: adverse events; OR: odds ratio; CI: confidence interval. |
| Discussion | ▴Top |
Our results showed that FMT significantly restored the normal microbiome and reduced the risk of gastrointestinal infections in HIV patients receiving antiretroviral therapy. Furthermore, AEs associated with FMT, such as distention and bloating, were comparable between HIV and health controls. In addition to GIT infections, disturbance of gut microbiota can also cause graft-versus-host disease (GVHD) in allo-hematopoietic stem cell transplantation (HSCT) patients.
In the FMT process, a healthy individual’s feces are mixed with saline or water and then filtered. The mixture is delivered through a nasogastric tube, nasojejunal tube, upper GIT endoscopy, colonoscopy, or enema [10, 31] Clostridium difficile is one of the common organisms causing diarrhea in immunocompromised patients.
Kelly et al [28] reported improvement of Clostridium difficile symptoms in 89% of patients after FMT within 12 weeks of follow-up. Most patients received FMT through lower GIT endoscopy. Because of the retrospective nature of this study, the reported side effects were incomplete [28]. A study also mentioned the impact of the route of administration of FMT. The upper GIT route carried a higher incidence of side effects than the lower GIT route [32, 33]. This is consistent with other systematic reviews and meta-analyses [33-35].
The variation in donor microbiota and the presence of unrecognized infectious agents can explain the occurrence of the side effects [36]. The mortality rate from FMT was low (0.13%) and may have occurred due to aspiration pneumonia [37]. Few adverse effects of FMT are reported and usually resolve spontaneously shortly after the procedure. Serious side effects of FMT are generally related to anesthesia or the endoscopic procedure [38]. The lack of accurate information about the route and dose of FMT makes the expansion of the procedure questionable [39].
Gathe Jr. et al [29] reported the failure of FMT to control diarrhea when taken from a relative. However, a specimen from a nonfamily member showed improvement in the symptoms. The choice of donor plays a role in the success of FMT [29]. Another case in 2013 showed symptoms improvement when treated with FMT from his mother after the antibiotic treatment failure [26].
Ott et al [18] studied the efficacy of a sterile fecal filtrate transfer (FFT) prepared from additional filtration of FMT. FFT contains antimicrobial compounds of bacterial origin (e.g., bacteriocins) or bacteriophages. FFT increases the success rate of FMT as it avoids the hazards of the transfer of living organisms [18].
Multiple doses of oral FMT also showed improvement in symptoms during 48 weeks of follow-up, compared to previous supplements [25]. Utay et al [24] also reported the safety and tolerability of repeated doses of oral FMT. However, they did not explain the required dose or duration. The small number of study participants limits the dependence on the results [24]. The study by Garcia-Garcia-de-Paredes et al reported the resolution of diarrhea in 87% of patients with Clostridium difficile infection. FMT was superior to the use of antibiotics that attack the bacteria as well as damage the intestinal microbiota [40].
Strength
To the best of our knowledge, no previous meta-analysis evaluated FMT efficacy in HIV patients. We included all types of studies reporting available results about FMT procedures in HIV patients. All of the included studies were conducted between 2013 and 2021.
Limitations
The limited number of randomized control trials weakens the meta-analysis’s evidence level. The included studies contain few patients, limiting the study of efficacy and possible side effects of the FMT procedure. Another limitation is that the included studies did not provide enough data about the presence of comorbidities, such as inflammatory bowel disease, ischemic heart disease, hypertension, and diabetes.
We recommend conducting more randomized trials with lagers number of patients. The type of fecal specimen donor should be studied.
Conclusions
Current evidence demonstrates that FMT is safe and effective in HIV patients suffering from alterations in gut microbiota. We recommend further multi-centric clinical studies to address the optimal transplant amount and source for FMT. To the best of our knowledge, this is the first meta-analysis to assess the safety and efficacy of FMT in patients with HIV.
| Supplementary Material | ▴Top |
Suppl 1. Newcastle-Ottawa quality assessment scale for cohort and case control studies.
Suppl 2. Funnel plot of included studies.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
Both authors have no conflict of interest.
Informed Consent
Not applicable.
Author Contributions
AM contributed to conceptualizing, database search, data collection, data analysis, writing, proofreading/editing the manuscript. MIM contributed to the database search, data collection, proofreading, and manuscript editing.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Mohapatra A, Sahoo, D. Review on HIV AIDS. Int J Psychosoc Rehabil. 2019;23:521-527.
doi - Avert The Science of HIV and AIDS - Overview Prof Resour. 2018;1-12.
- Mehandru S, Tenner-Racz K, Racz P, Markowitz M. The gastrointestinal tract is critical to the pathogenesis of acute HIV-1 infection. J Allergy Clin Immunol. 2005;116(2):419-422.
doi pubmed - Ng Chee Ping. 基因的改变NIH Public Access. Bone 2013;23:1-7.
- Serlin MH, Dieterich D. Gastrointestinal disorders in HIV. Glob HIV/AIDS Med. 2008;251-260.
doi - Brenchley JM, Douek DC. HIV infection and the gastrointestinal immune system. Mucosal Immunol. 2008;1(1):23-30.
doi pubmed pmc - Bhaijee F, Subramony C, Tang SJ, Pepper DJ. Human immunodeficiency virus-associated gastrointestinal disease: common endoscopic biopsy diagnoses. Patholog Res Int. 2011;2011:247923.
doi pubmed pmc - Lin L, Zhang J. Role of intestinal microbiota and metabolites on gut homeostasis and human diseases. BMC Immunol. 2017;18(1):2.
doi pubmed pmc - Rashighi M, Harris JE. 乳鼠心肌提取 HHS Public Access. Physiol Behav. 2017;176:139-148.
doi - Ouyang J, Isnard S, Lin J, Fombuena B, Peng X, Nair Parvathy S, Chen Y, et al. Treating from the inside out: relevance of fecal microbiota transplantation to counteract gut damage in GVHD and HIV infection. Front Med (Lausanne). 2020;7:421.
doi pubmed pmc - Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. PLoS Med. 2021;18(3):e1003583.
doi pubmed pmc - Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane Handbook for Systematic Reviews of Interventions. John Wiley & Sons. 2019.
- Wells GA, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Oxford. 2000.
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-560.
doi pubmed pmc - Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50(4):1088-1101.
pubmed - Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;56(2):455-463.
doi pubmed - Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629-634.
doi pubmed pmc - Ott SJ, Waetzig GH, Rehman A, Moltzau-Anderson J, Bharti R, Grasis JA, Cassidy L, et al. Efficacy of sterile fecal filtrate transfer for treating patients with clostridium difficile infection. Gastroenterology. 2017;152(4):799-811.e797.
doi pubmed - Mandalia A, Ward A, Tauxe W, Kraft CS, Dhere T. Fecal transplant is as effective and safe in immunocompromised as non-immunocompromised patients for Clostridium difficile. Int J Colorectal Dis. 2016;31(5):1059-1060.
doi pubmed pmc - Ling Z, Jin C, Xie T, Cheng Y, Li L, Wu N. Alterations in the fecal microbiota of patients with HIV-1 infection: an observational study in a Chinese population. Sci Rep. 2016;6:30673.
doi pubmed pmc - Shogbesan O, Poudel DR, Victor S, Jehangir A, Fadahunsi O, Shogbesan G, Donato A. A systematic review of the efficacy and safety of fecal microbiota transplant for clostridium difficile infection in immunocompromised patients. Can J Gastroenterol Hepatol. 2018;2018:1394379.
doi pubmed pmc - Elopre L. Division of infectious diseases. Ann Intern Med. 2013;229:779-780.
doi - Zhou Y, Ou Z, Tang X, Zhou Y, Xu H, Wang X, Li K, et al. Alterations in the gut microbiota of patients with acquired immune deficiency syndrome. J Cell Mol Med. 2018;22(4):2263-2271.
doi pubmed pmc - Utay NS, Monczor AN, Somasunderam A, Lupo S, Jiang ZD, Alexander AS, Finkelman M, et al. Evaluation of six weekly oral fecal microbiota transplants in people with HIV. Pathog Immun. 2020;5(1):364-381.
doi pubmed pmc - Serrano-Villar S, Talavera-Rodriguez A, Gosalbes MJ, Madrid N, Perez-Molina JA, Elliott RJ, Navia B, et al. Fecal microbiota transplantation in HIV: A pilot placebo-controlled study. Nat Commun. 2021;12(1):1139.
doi pubmed pmc - Klis S, van der Werf TS, Phillips RO, Sarfo FS, Wansbrough-Jones M, Stienstra Y. Oral treatment for patients with Buruli ulcer co-infected with HIV: think twice. AIDS. 2014;28(5):797-798.
doi pubmed - Schunemann M, Oette M. Fecal microbiota transplantation for Clostridium difficile-associated colitis in a severely immunocompromized critically ill AIDS patient: a case report. AIDS. 2014;28(5):798-799.
doi pubmed - Kelly CR, Ihunnah C, Fischer M, Khoruts A, Surawicz C, Afzali A, Aroniadis O, et al. Fecal microbiota transplant for treatment of Clostridium difficile infection in immunocompromised patients. Am J Gastroenterol. 2014;109(7):1065-1071.
doi pubmed pmc - Gathe JC, Jr., Diejomaoh EM, Mayberry CC, Clemmons JB. Fecal transplantation for Clostridium difficile-"All Stool May Not Be Created Equal". J Int Assoc Provid AIDS Care. 2016;15(2):107-108.
doi pubmed - Elopre L, Rodriguez M. Fecal microbiota therapy for recurrent Clostridium difficile infection in HIV-infected persons. Ann Intern Med. 2013;158(10):779-780.
doi pubmed pmc - Gupta S, Allen-Vercoe E, Petrof EO. Fecal microbiota transplantation: in perspective. Therap Adv Gastroenterol. 2016;9(2):229-239.
doi pubmed pmc - Furuya-Kanamori L, Doi SA, Paterson DL, Helms SK, Yakob L, McKenzie SJ, Garborg K, et al. Upper versus lower gastrointestinal delivery for transplantation of fecal microbiota in recurrent or refractory clostridium difficile infection: a collaborative analysis of individual patient data from 14 studies. J Clin Gastroenterol. 2017;51(2):145-150.
doi pubmed - Tariq R, Syed T, Yadav D, Prokop LJ, Singh S, Loftus EV, Jr., Pardi DS, et al. Outcomes of fecal microbiota transplantation for c. difficile infection in inflammatory bowel disease: a systematic review and meta-analysis. J Clin Gastroenterol. 2023;57(3):285-293.
doi pubmed - Quraishi MN, Widlak M, Bhala N, Moore D, Price M, Sharma N, Iqbal TH. Systematic review with meta-analysis: the efficacy of faecal microbiota transplantation for the treatment of recurrent and refractory Clostridium difficile infection. Aliment Pharmacol Ther. 2017;46(5):479-493.
doi pubmed - Singh T, Bedi P, Bumrah K, Gandhi D, Arora T, Verma N, Schleicher M, et al. Fecal microbiota transplantation and medical therapy for clostridium difficile infection: meta-analysis of randomized controlled trials. J Clin Gastroenterol. 2022;56(10):881-888.
doi pubmed - Wang S, Xu M, Wang W, Cao X, Piao M, Khan S, Yan F, et al. Systematic review: adverse events of fecal microbiota transplantation. PLoS One. 2016;11(8):e0161174.
doi pubmed pmc - Marcella C, Cui B, Kelly CR, Ianiro G, Cammarota G, Zhang F. Systematic review: the global incidence of faecal microbiota transplantation-related adverse events from 2000 to 2020. Aliment Pharmacol Ther. 2021;53(1):33-42.
doi pubmed - Terra DAA, Vilela EG, Silva ROS, LeAo LA, Lima KS, Passos R, Diniz AN, et al. Structuring a fecal microbiota transplantation center in a university hospital in Brazil. Arq Gastroenterol. 2020;57(4):434-458.
doi pubmed - Wortelboer K, Nieuwdorp M, Herrema H. Fecal microbiota transplantation beyond Clostridioides difficile infections. EBioMedicine. 2019;44:716-729.
doi pubmed pmc - Garcia-Garcia-de-Paredes A, Rodriguez-de-Santiago E, Aguilera-Castro L, Ferre-Aracil C, Lopez-Sanroman A. [Fecal microbiota transplantation]. Gastroenterol Hepatol. 2015;38(3):123-134.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Gastroenterology Research is published by Elmer Press Inc.


