| Gastroenterology Research, ISSN 1918-2805 print, 1918-2813 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Gastroenterol Res and Elmer Press Inc |
| Journal website http://www.gastrores.org |
Case Report
Volume 3, Number 3, June 2010, pages 134-138
Actinomycosis of the Pancreas: A Case Report and Review
Amalanshu Jhaa, f, Anusak Yiengpruksawanb, Arthur A. Christianoc, Neeti Jhad, Prajna Latikae
aDepartment of Surgery, Bronx-Lebanon Hospital Center, Bronx, New York, USA
bSurgical Oncology, Minimal Invasive and Robotic Surgery, The Valley Hospital, Ridgewood, New Jersey, USA
cDepartment of Pathology, The Valley Hospital, Ridgewood, New Jersey, USA
dDepartment of Internal Medicine, Lincoln Medical and Mental Health Center, Bronx, New York, USA
eDepartment of Internal Medicine, Bronx-Lebanon Hospital Center, Bronx, New York, USA
fCorresponding author: Department of Surgery, Bronx-Lebanon Hospital Center, 98 Johnson Rd, Scarsdale, NY 10583, USA
Manuscript accepted for publication May 28, 2010
Short title: Actinomycosis of Pancreas
doi: https://doi.org/10.4021/gr2010.05.207w
| Abstract | ▴Top |
Actinomyces is a normal commensal of the upper aerodigestive tract, colon and female reproductive tract. It can give rise to invasive disease in case of any breach in mucosal integrity, as well as, in patients with immunosuppression. Rarely, actinomycosis can involve the pancreas especially after episodes of pancreatitis or in post operative patients. We observed a case of actinomycosis affecting recurrent intraductal papillary mucinous neoplasm (IPMN) of pancreatic remnant, 5 years after a Whipple's procedure. Our patient, a 66 years old male with a history of Whipple's procedure for IPMN of pancreatic uncinate process, presented with repeated episodes of acute pancreatitis. Repeated radiological investigations (CT, MRI and EUS) revealed resolving pancreatitis with recurrent IPMN of the pancreatic tail. The patient underwent laparobotic assisted resection of the remnant pancreas and spleen 3 months later. Intraoperatively, in addition to the recurrent IPMN of pancreatic tail, we found a dense peripancreatic desmoplastic reaction with areas of thick yellow pus pockets in the remnant pancreatic body. Bacteriology and histopathology revealed it as a recurrent IPMN associated with actinomycosis of pancreas with chronic xanthogranulomatous changes. We conclude that actinomycosis of the pancreas is a rare entity with only 5 cases reported in English literature to the best of our knowledge. If diagnosed preoperatively, early institution of antibiotics can improve the surgical outcome. Fortunately, after diagnosis, we were able to start antibiotics in early postoperative period with successful outcome.
Keywords: Pancreas; Actinomycosis; Intraductal papillary mucinous tumor (IPMN)
| Introduction | ▴Top |
Actinomyces is a normal commensal of the upper aerodigestive tract, colon and female reproductive tract. It can give rise to invasive disease after a breach of mucosal integrity, as well as, in patients with immunosuppression [1-6].
It causes an indolent, fibrosing infection of any organ, with formation of sinuses discharging pus with ‘sulfur granules’. It has been shown to form internal fistulae, mucosal/skin ulcers [7] and occasionally an inflammatory mass which can mimic a malignancy [8, 9].
Actinomycotic involvement of the pancreas is extremely rare. We report a case of actinomycosis of the pancreatic remnant having recurrent intraductal papillary mucinous neoplasm (IPMN). Our patient had pancreaticoduodenectomy 5 years ago for IPMN of the pancreas. Subsequently, he had several episodes of pancreatitis before being diagnosed with recurrent IPMN of pancreas. Actinomycosis was reported in the resected pancreas and the splenic hilum.
| Case Report | ▴Top |
A 66 years old man presented with recurrent epigastric pain. Computed tomography (CT) showed a 3 cm cystic lesion in the uncinate process of the pancreas. Endoscopic ultrasonography (EUS), further, revealed a 2.6 cm x 1.6 cm cyst in the uncinate process with an irregular wall and papillary growth.
He underwent a Whipple's procedure at a different hospital. The cyst was found to be an IPMN. His postoperative course was stormy with pancreatitis of the pancreatic remnant, pulmonary embolism necessitating an IVC filter placement and prolonged hospitalization (2 months).
Five years later he presented with symptoms of acute pancreatitis. A CT scan showed pancreatitis with a 15 mm acute fluid collection within the pancreatic remnant (Fig. 1).
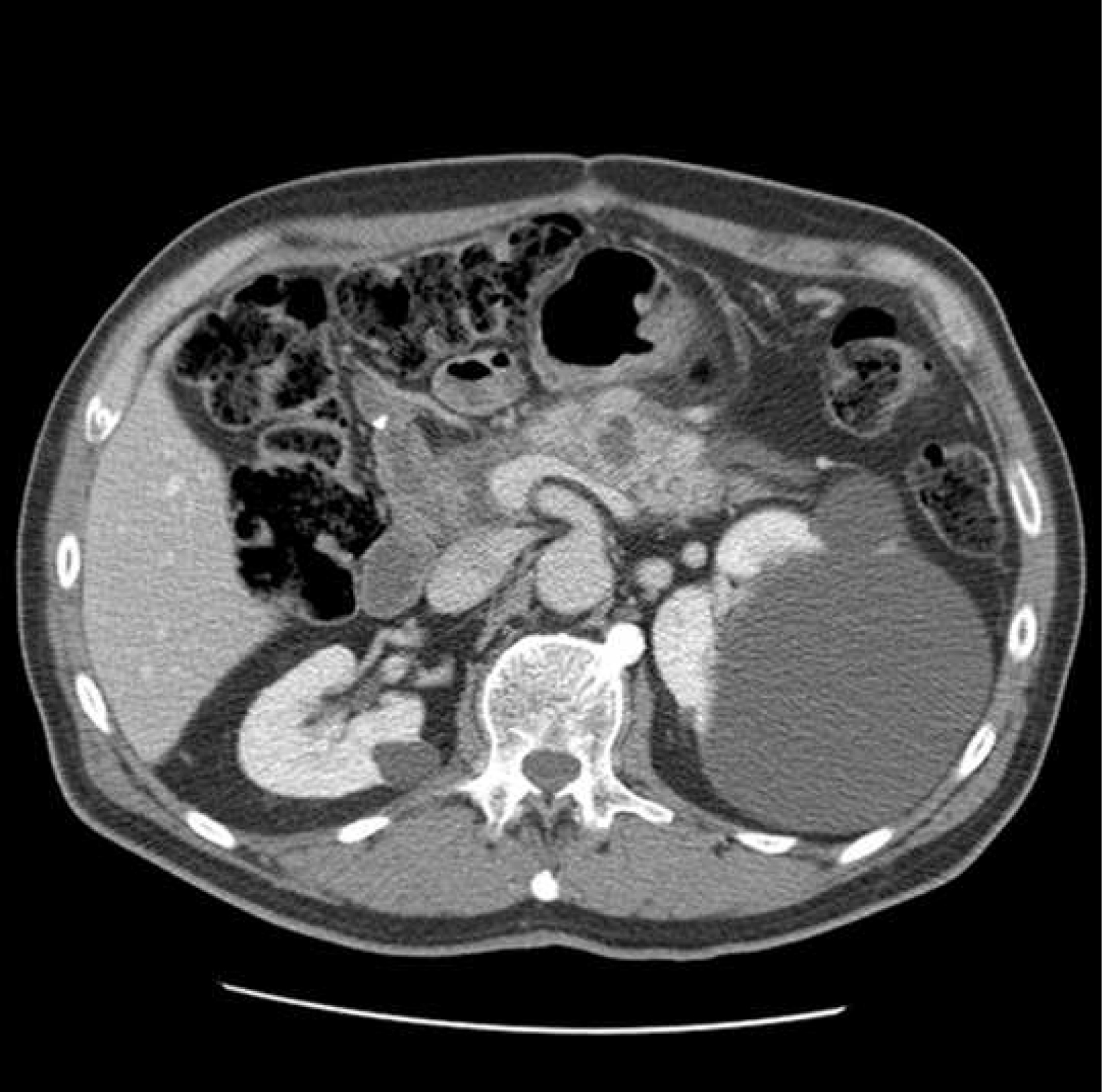 Click for large image | Figure 1. Acute pancreatitis with fluid collection. |
The patient had an uneventful recovery from pancreatitis. He continued to have episodes of epigastric pain, malaise and weight loss over next 5 months. Follow up Magnetic resonance cholangiopancreatography (MRCP) two months after pancreatitis showed resolution of acute fluid collection. It also revealed a new cystic lesion in the pancreatic tail (Fig. 2) with stenosis of the main pancreatic duct and distal dilatation (Fig. 3). Further evaluation by EUS showed a 22 mm x 24 mm cyst in the pancreatic tail.
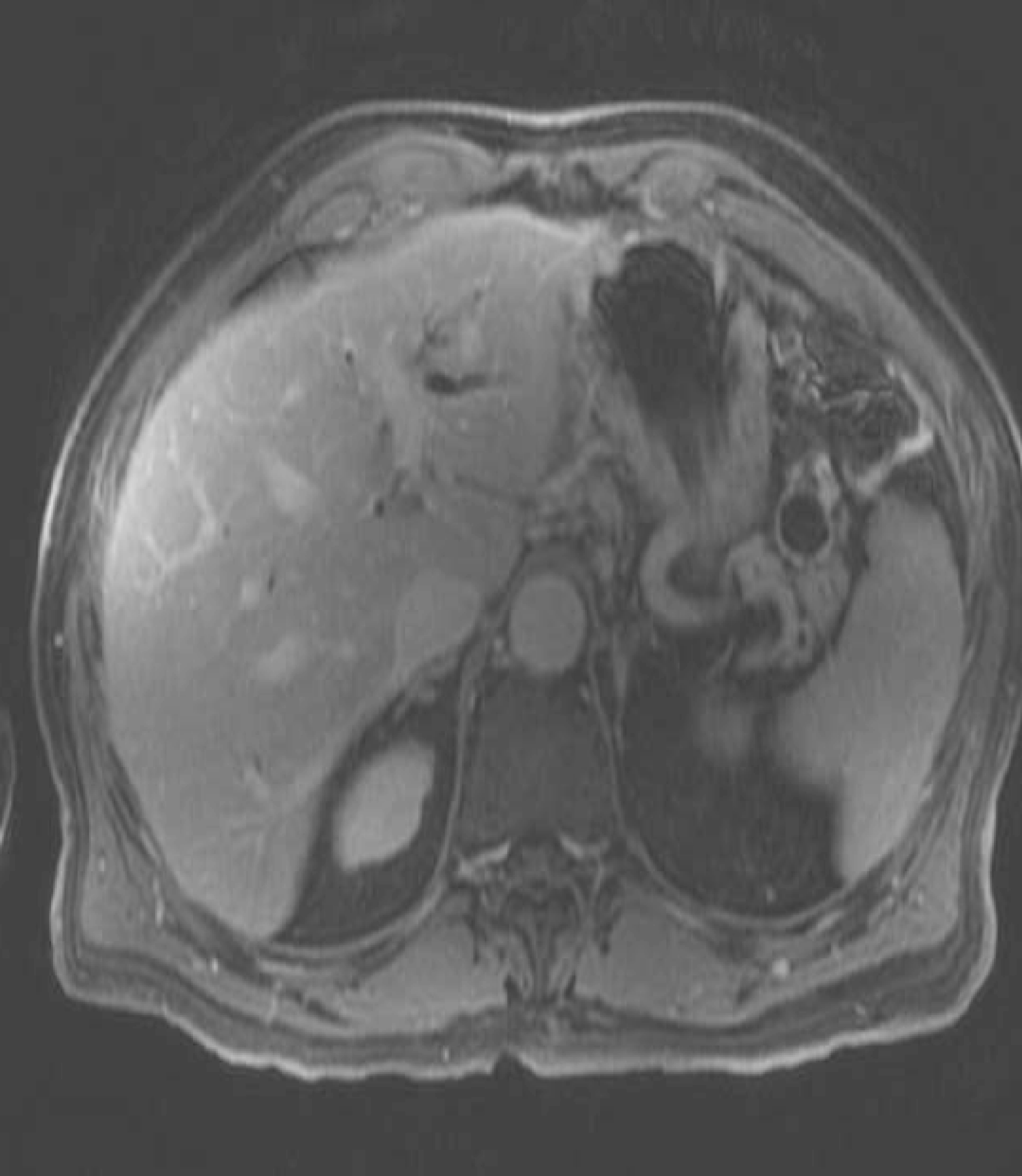 Click for large image | Figure 2. Recurrent IPMN cyst of pancreatic tail. |
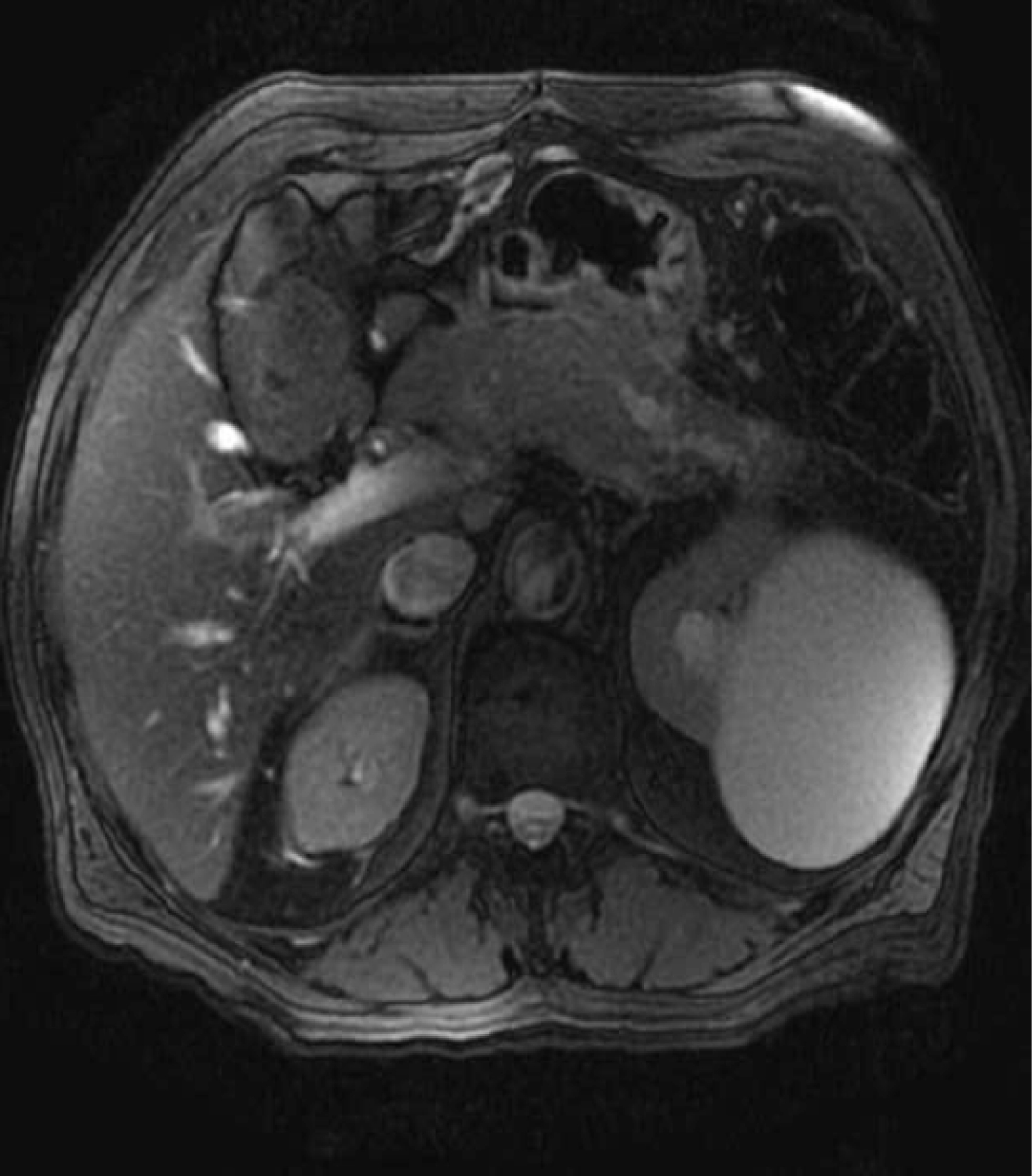 Click for large image | Figure 3. Pancreatic duct stenosis with distal dilatation. |
The patient underwent laparobotic assisted pancreaticosplenectomy 3 months later. Intraoperative findings included a cyst (recurrent IPMN on frozen section) in the pancreatic tail. Additionally, there was extensive fibrosis around the pancreas with adhesions to the surrounding structures including the stomach and the splenic flexure of colon. Several yellow pus pockets were found in the pancreatic tail extending to the splenic hilum (Fig. 4).
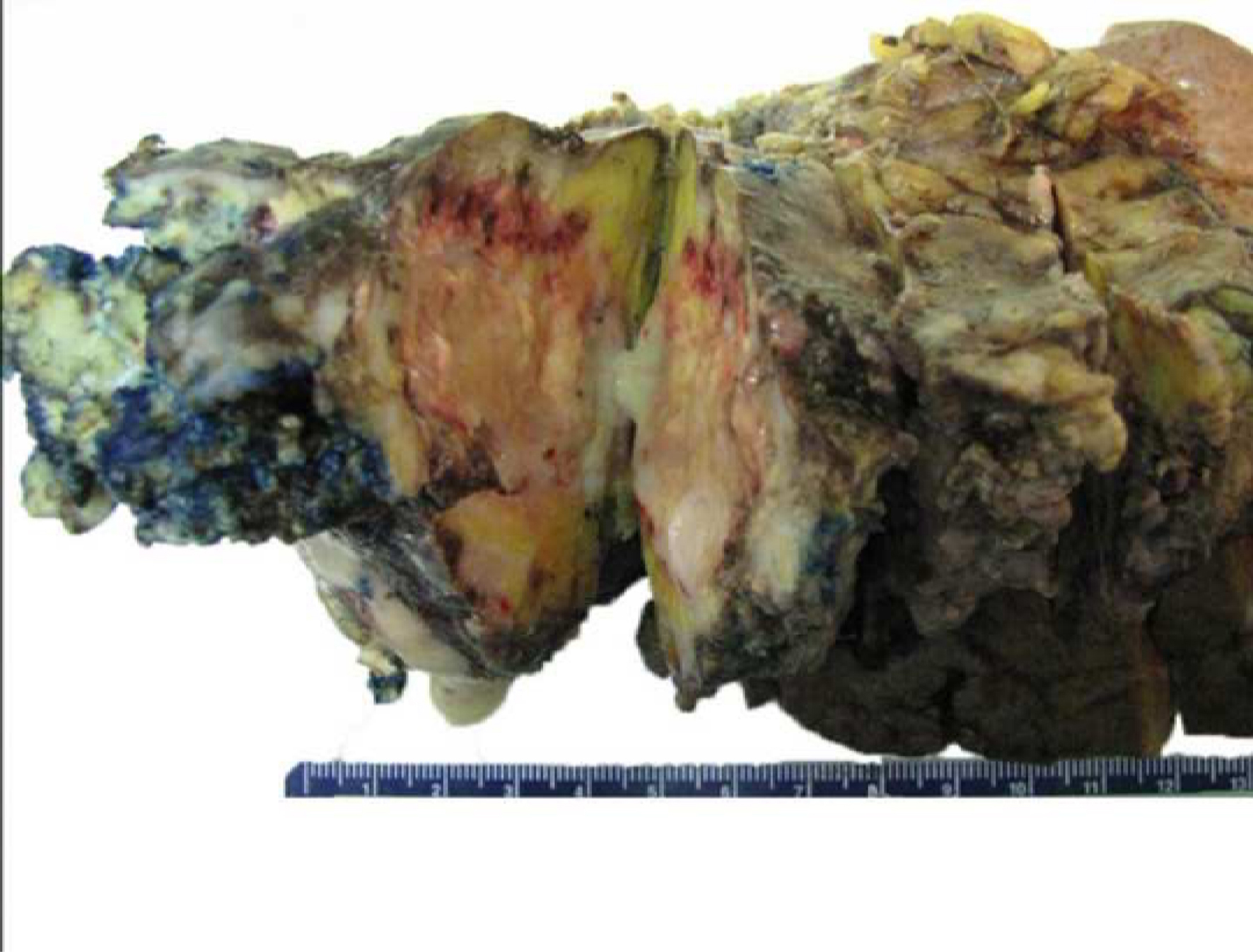 Click for large image | Figure 4. Gross specimen of resected remnant pancreas with spleen. |
Histopathology revealed recurrent IPMN with colonies of actinomyces in the pancreatic tissue in a background of inflammatory infiltrate and foamy histiocytes - suggestive of chronic xanthogranulomatous pancreatitis (Fig. 5).
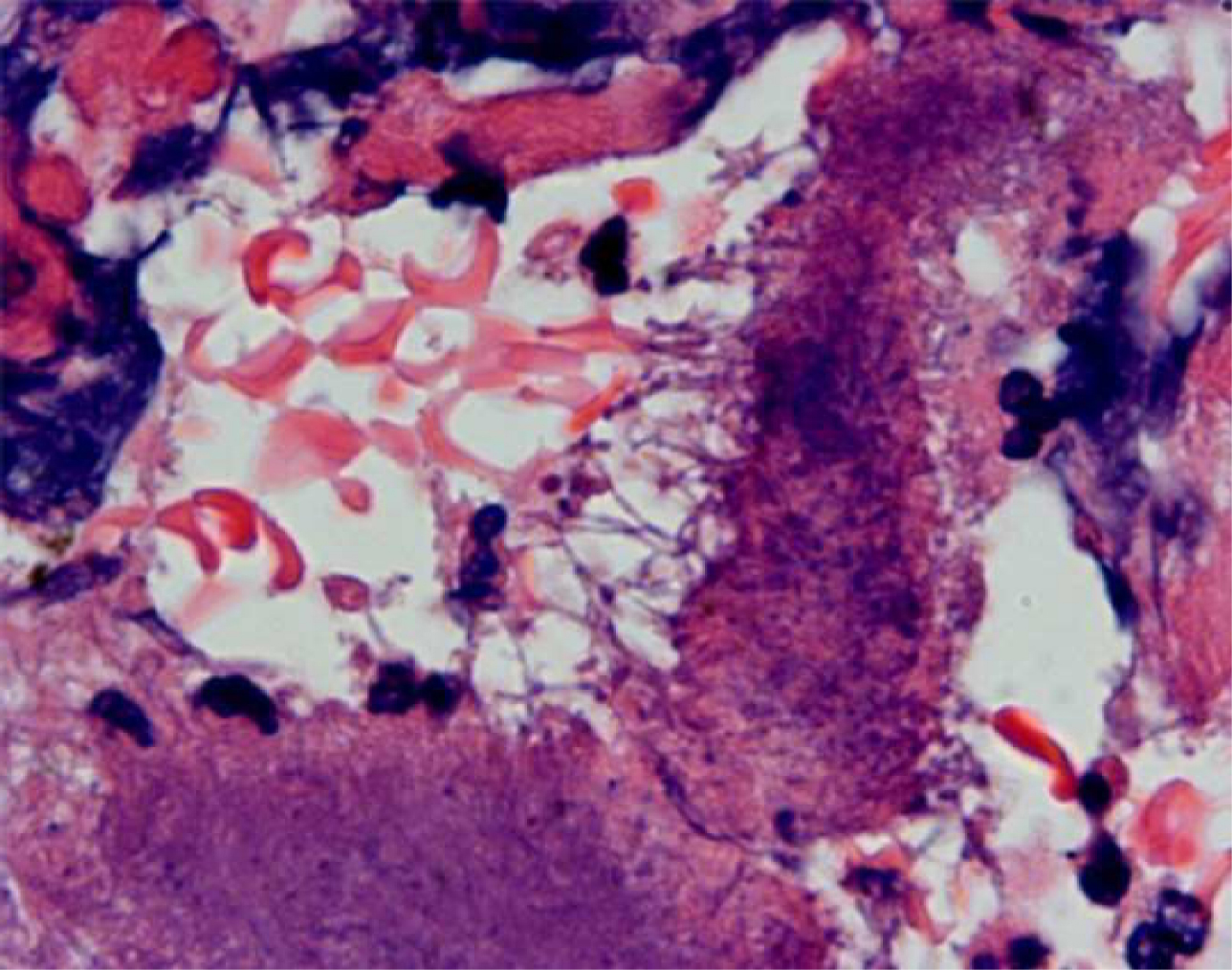 Click for large image | Figure 5. Branching bacterial filaments at the edge of sulfur granule. Note surrounding inflammation. |
The patient was treated with intravenous penicillin for 4 to 6 weeks followed by oral amoxicillin for further 6 to 8 months. The patient is healthy at 2 years of follow-up.
| Discussion | ▴Top |
Etiology and pathogenesis
Actinomycosis is a rare opportunistic disease usually caused by the species Actinomyces israelii. Other species in the genus Actinomyces which may cause human infections include: A. odontolyticus, A. naeslundii, A. meyeri, A. gerencseriae and A. viscosus [10]. These organisms were once believed to be fungi due to their branching filamentous morphology. They lack a nuclear membrane; their cell walls lack chitin and are affected by penicillin but not by antifungal agents [11]. Hence, they are now considered to be higher forms of prokaryotic bacteria.
They are gram positive filamentous bacteria. They are typically anaerobic or microaerophilic, hence surviving as commensal in oral cavity, gastrointestinal tract and female genital tract. In fact, oral colonization has been shown to occur in 97% of infants by 2 years of age [12]. No known natural source or natural habitat is known besides human beings and animals.
They can acquire pathogenicity after invasion of tissues. They are known to enter tissues with any mucosal breach due to dental procedures, oral surgery [3], after inflammatory diseases of gastrointestinal tract including diverticulitis, appendicitis and intestinal perforation, and post surgery [4-6] or long standing intrauterine contraceptive devices [2]. Foreign bodies such as fish bone [1], IUCD's [2] have been reported to facilitate infection.
Infection is usually chronic and slowly progressive. Due to its infiltrating nature, it causes a dense desmoplastic reaction in the surrounding tissue. It slowly grows by contiguous spread ignoring anatomical planes. With further growth, the center of the infection site liquefies to become purulent material.
Indurated lesions are more apparent in abdominal or cervicofacial locations as compared with pulmonary or central nervous system (CNS) lesions. Sinus tracts might form to the skin or surrounding viscera due to its infiltrative nature.
Microscopically, ‘sulfur granules’ are the hallmark of the disease. They consist of a conglomeration of filamentous bacteria. The filaments are better visualized at the margins of these granules. The surrounding tissues show dense inflammatory infiltrates of neutrophils, lymphocytes and foamy macrophages.
Clinical presentation
Actinomycosis can involve any tissue of the body, but clinical patterns can be classified broadly into 6 categories in the order of frequency: Oro-cervicofacial (55%) [4], abdominopelvic (20%) [4, 11], thoracic (15%) [13], rarely CNS, musculoskeletal and disseminated actinomycosis.
In oral and cervicofacial region, actinomycosis can present as a soft tissue swelling, abscess, sinus, mass [4] or an ulcer [7]. Although any part of oral cavity or neck can be involved, the classic location is described at the angle of mandible [14].
In the thorax, the disease commonly presents as a pulmonary mass with ‘air bronchogram’ (patent bronchus in a mass, also called ‘open bronchus sign’) [15], pneumonia, empyema or cavitations (usually multiple) [15]. Mediastinal disease, although rare, commonly involves pericardium [16].
Rarely, the CNS is involved. Brain abscess is the most common presentation with CT showing ring-enhancing lesions.
Abdominal actinomycosis commonly presents as a slowly growing mass mimicking malignancy or as an abscess. It can involve any site in the abdominal cavity but it is more frequent in the ileocecal region [17]. Other areas reported to be involved include liver [18], gallbladder [19], pelvis (perianal and perirectal disease) and the retroperitoneum including ureters and kidneys [20].
Pancreatic involvement is extremely uncommon. It has been reported only 5 times in the past to the best of our knowledge (Table 1). The earliest report was by Parsons et al [21] in 1929 of a fatal case of pancreatic actinomycosis, initially suspected as pancreatic necrosis. In all the other case reports, the initial suspicion was that of pancreatic malignancy due to their presentation as a pancreatic mass. Two patients had chronic stent associated pancreatic actinomycosis [18] and three were linked to prior surgery [8, 9, 22]. Chronic pancreatitis was associated with actinomycosis in 3 patients [8, 18], while prior pancreatic surgery was present in 2 patients [8, 9] and one patient had both prior surgery and pancreatitis [8] as in our patient.
 Click to view | Table 1. Reported Cases of Pancreatic Actinomycosis in Literature |
We believe that our patient had the seeding of pancreas by actinomyces by repeated episodes of pancreatitis aided by reflux of gastrointestinal contents via the anastomosis of remnant pancreatic duct to jejunum.
In a symptomatic patient who had a prior surgery and also who had recurrent episodes of pancreatitis, it is not unreasonable to consider actinomycosis of the pancreas in the differential diagnosis. A CT or EUS guided FNA of inflammatory collection around pancreas may yield the diagnosis of actinomycosis [8] which can then be successfully treated by antibiotics. This may improve the surgical outcome in elective or semi-emergent cases.
Financial Support
We did not have any financial support or grant for the work. We do not have any disclosures or any product endorsements.
Conflict of Interest
We declare that we have no conflict of interests for this manuscript.
| References | ▴Top |
- Yamada H, Kondo S, Kamiya J, Nagino M, Miyachi M, Kanai M, Hayata A,
et al . Computed tomographic demonstration of a fish bone in abdominal actinomycosis: report of a case. Surg Today. 2006;36(2):187-189.
pubmed doi - Russo TA: Actinomycosis. In: Fauci AS, Braunwald E, Isselbacher KJ et al. (eds): Harrison’s Principles of Internal Medicine, 14th ed. New York: McGraw-Hill, 1998, pp 989-991.
- Zitsch RP
3rd , Bothwell M. Actinomycosis: a potential complication of head and neck surgery. Am J Otolaryngol. 1999;20(4):260-262.
pubmed doi - Bennhoff DF. Actinomycosis: diagnostic and therapeutic considerations and a review of 32 cases. Laryngoscope. 1984;94(9):1198-1217.
pubmed - Weese WC, Smith IM. A study of 57 cases of actinomycosis over a 36-year period. A diagnostic 'failure' with good prognosis after treatment. Arch Intern Med. 1975;135(12):1562-1568.
pubmed doi - Harvey JC, Cantrell JR, Fisher AM. Actinomycosis: its recognition and treatment. Ann Intern Med. 1957;46(5):868-885.
pubmed - Alamillos-Granados FJ, Dean-Ferrer A, Garcia-Lopez A, Lopez-Rubio F. Actinomycotic ulcer of the oral mucosa: an unusual presentation of oral actinomycosis. Br J Oral Maxillofac Surg. 2000;38(2):121-123.
pubmed doi - Somsouk M, Shergill AK, Grenert JP, Harris H, Cello JP, Shah JN. Actinomycosis mimicking a pancreatic head neoplasm diagnosed by EUS-guided FNA. Gastrointest Endosc. 2008;68(1):186-187.
pubmed doi - Hsu JT, Lo HC, Jan YY, Chen HM. Actinomycosis mimicking recurrent carcinoma after Whipple's operation. World J Gastroenterol. 2005;11(11):1722-1724.
pubmed - Schaal KP, Lee HJ. Actinomycete infections in humans—a review. Gene. 1992;115(1-2):201-211.
pubmed doi - Russo TA. Agents of Actinomycosis. In: Mandell, Bennett & Dolin: Principles of Infectious Diseases, 6th ed. Churchill Livingstone, chap - 253.
- Sarkonen N, Kononen E, Summanen P, Kanervo A, Takala A, Jousimies-Somer H. Oral colonization with Actinomyces species in infants by two years of age. J Dent Res. 2000;79(3):864-867.
pubmed doi - Bates M, Cruickshank G. Thoracic actinomycosis. Thorax. 1957;12(2):99-124.
pubmed doi - Samuels RH, Martin MV. A clinical and microbiological study of Actinomycetes in oral and cervicofacial lesions. Br J Oral Maxillofac Surg. 1988;26(6):458-463.
pubmed doi - Flynn MW, Felson B. The roentgen manifestations of thoracic actinomycosis. Am J Roentgenol Radium Ther Nucl Med. 1970;110(4):707-716.
pubmed - Fife TD, Finegold SM, Grennan T. Pericardial actinomycosis: case report and review. Rev Infect Dis. 1991;13(1):120-126.
pubmed - Smego RA
Jr , Foglia G. Actinomycosis. Clin Infect Dis. 1998;26(6):1255-1261, quiz 1262-1253.
pubmed - Harsch IA, Benninger J, Niedobitek G, Schindler G, Schneider HT, Hahn EG, Nusko G. Abdominal actinomycosis: complication of endoscopic stenting in chronic pancreatitis?. Endoscopy. 2001;33(12):1065-1069.
pubmed doi - Merle-Melet M, Mory F, Stempfel B, Maurer P, Regent D, Parent S, Tortuyaux JM,
et al . Actinomyces naeslundii, acute cholecystitis, and carcinoma of the gallbladder. Am J Gastroenterol. 1995;90(9):1530-1531.
pubmed - Khalaff H, Srigley JR, Klotz LH. Recognition of renal actinomycosis: nephrectomy can be avoided. Report of a case. Can J Surg. 1995;38(1):77-79.
pubmed - Parsons HH. Actinomycosis of Pancreas: Report of Case. Cal West Med. 1931;35(6):452.
pubmed - Halevy A, Blenkharn JI, Christodoloupolous J, Blumgart LH. Actinomycosis of the pancreas. Br J Surg. 1987;74(2):150.
pubmed doi
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Gastroenterology Research is published by Elmer Press Inc.


